We don’t usually question how a drug received FDA approval; we just assume that people in power know what they’re doing and have the public’s best interests at heart, right? The FDA, which oversees the safety of medications in the United States, effectively controls $2.7 trillion of the country’s economy. The FDA often publishes warning letters about the safety of medicines on the market, whether over-the-counter or prescribed by a doctor. According to a new analysis, these warnings are now at their lowest since 2008. That isn’t particularly concerning until you consider that a third of the drugs approved for sale by the FDA had safety concerns issued later on. To make matters worse, everyone affected by unsafe medications encountered significant medical consequences, often far-reaching and unhealable, ranging from carcinomas, birth deformities, organ failure, and even death, to realize that our trust should not be given so easily.
Today, we’ll look at some of history’s most notable and impactful drug recalls. Here are 25 FDA-Approved drugs that eventually got banned.

ZOFRAN, 2011
 https://www.drugwatch.com/zofran/
https://www.drugwatch.com/zofran/ In 1991, Zofran was approved as an anti-nausea medicine for cancer chemotherapy patients. Because of its efficacy, doctors everywhere soon began prescribing Zofran to pregnant women to alleviate the effects of morning sickness. Although the FDA did not specifically allow this use, Zofran swiftly became one of the most commonly prescribed medications in the United States, if not the world. The FDA recalled the 32 mg intravenous dosage of Zofran in 2012. According to studies, it poses a significant risk to the heart.
To make matters worse, women who took Zofran for morning sickness had children with devastating heart abnormalities. Many families blamed Zofran for these side effects. According to the lawsuits, GSK advertised Zofran for this “off-label” use while being aware of the harm to fetuses. These claims were eventually settled for billions of dollars.
EFALIZUMAB (RAPTIVA), 2009
 https://www.ema.europa.eu/en/news/eu-wide-recall-raptiva-efalizumab-be-initiated#:~:text=The%20European%20Medicines%20Agency%20has,from%20wholesalers%2C%20pharmacies%20and%20hospitals.
https://www.ema.europa.eu/en/news/eu-wide-recall-raptiva-efalizumab-be-initiated#:~:text=The%20European%20Medicines%20Agency%20has,from%20wholesalers%2C%20pharmacies%20and%20hospitals. Raptiva was on the market for six years before being discontinued in 2009. The medication was used to treat psoriasis, characterized by elevated silvery flaking of the skin in adults. The drug was eventually recalled after it was determined that the medicine might trigger progressive multifocal leukoencephalopathy or PML. This rare and usually fatal condition causes inflammation or progressive damage to the brain’s central nervous system and white matter. Raptiva had been used to treat around 46,000 patients worldwide after its FDA approval in 2003, with roughly 2,000 patients in the United States receiving it at the time of its recall.
BELVIQ (LORCASERIN), 2020
 https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-withdrawal-weight-loss-drug-belviq-belviq-xr-lorcaserin-market
https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-withdrawal-weight-loss-drug-belviq-belviq-xr-lorcaserin-market We know we may encounter risks when using a prescription drug to lose some extra weight. Sometimes the weight loss is enough to weather the resultant bad news. Belviq, a prescription weight loss medication, was recently removed from the market. A clinical trial revealed an increase in the occurrence of cancer within five years in those who had used the drug. As such, the FDA recommended that Belviq’s manufacturer voluntarily remove it from the US market in February 2020.
TERFENADINE (SELDANE), 1998
 https://www.medicinenet.com/seldane_removed/views.htm
https://www.medicinenet.com/seldane_removed/views.htm This antihistamine was on the market for 13 years before it was recalled due to catastrophic heart complications that developed when it was used with either erythromycin or ketoconazole. Although the medicine was not considered to be an immediate threat, it was only eventually pulled because its manufacturer, Hoechst Marion Roussel (now Sanofi-Aventis), also sold fexofenadine (Allegra and Allegra-D), which the FDA assessed to be a far safer alternative. So it remained on the shelves. Until the manufacturer had a safer alternative. Shocking.
ZANTAC (RANITIDINE), 2020
 https://www.westvirginiapersonalinjurylawyer.net/fda-recalls-zantac-what-you-need-to-know/#Ranitidine_Effects
https://www.westvirginiapersonalinjurylawyer.net/fda-recalls-zantac-what-you-need-to-know/#Ranitidine_Effects In April 2020, the FDA recommended the recall of Zantac, a once popular over-the-counter drug for stomach ailments such as heartburn and acid reflux. It was recalled after testing revealed that it contained significant quantities of the carcinogen N-nitrosodimethylamine (NDMA), which causes a multitude of serious side effects. Ranitidine, in addition to inducing dizziness and gastrointestinal distress, has also been suspected to play a role in the development of cancer. Because this is an emerging issue, lawsuits are flowing in. Hundreds of personal injury cases have already been filed.
SIBUTRAMINE (MERIDIA), 2010
 https://www.oshmanlaw.com/sibutramine/#:~:text=Sibutramine%20Product%20Recall&text=In%20October%202010%2C%20the%20FDA,avoid%20all%20sibutramine%2Dcontaining%20products.
https://www.oshmanlaw.com/sibutramine/#:~:text=Sibutramine%20Product%20Recall&text=In%20October%202010%2C%20the%20FDA,avoid%20all%20sibutramine%2Dcontaining%20products. Meridia, an appetite suppressant, was recalled by the FDA in 2010 due to a 16% increased risk of having heart problems such as irregular heartbeat, non-fatal stroke, and cardiac arrest. Abbott Laboratories’ obesity “wonder drug” was on the market for 13 years and was often prescribed to those with a BMI of 30 or higher. In testimony before a Senate committee on September 30, 2004, FDA reviewer Dr David Graham listed Meridia with Crestor, Accutane, Bextra, and Serevent as drugs whose sales should be discontinued due to their deadly side effects, even going so far as to call Meridia “another Vioxx.” A deadly drug we’ll get to shortly.
ISOTRETINOIN (ACCUTANE), 2009
 https://www.aboutlawsuits.com/accutane-side-effects-ulcerative-colitis-9486/
https://www.aboutlawsuits.com/accutane-side-effects-ulcerative-colitis-9486/ Everyone growing up in the ’90s has probably heard of Accutane. Accutane was the go-to remedy to treat severe acne. It was used by about 13 million people worldwide between 1982 and 2009, when it was pulled off the market. However, generic Isotretinoin is still accessible from a variety of manufacturers. The medicine, made by Hoffmann-La Roche Inc., was found to cause serious negative side effects when used by pregnant women, such as birth deformities. It could also lead to other issues such as depression, psychosis, and even inflammatory bowel disease. Nosebleeds, joint pain, and skin rashes all eventually made their way to the list of negative side effects. Over 7,000 cases have been filed against Hoffman-La Roche since, with three verdicts totalling over $10 million.
LEVAMISOLE (ERGAMISOL), 2000
 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3498128/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3498128/ Every drug has its highs and its lows. Some evidently have more highs. Levamisole was prescribed for people experiencing worm infestations, rheumatoid arthritis, colon cancer, and breast cancer. Sounds like a noble drug, right? Unfortunately, apart from being mixed into street cocaine today to increase its effects, it was recalled because it caused neutropenia, agranulocytosis, and thrombotic vasculopathy, which resulted in retiform purpura—a purple eye discoloration that necessitated surgery. It is worth noting that Levamisole is still used to treat worms in animals in the United States today.
PALLADONE, 2005
 https://www.nbcnews.com/health/health-news/fda-halts-sales-painkiller-palladone-flna1c9441709
https://www.nbcnews.com/health/health-news/fda-halts-sales-painkiller-palladone-flna1c9441709 Palladone, a highly effective narcotic pain reliever, was recalled in 2005 after only a year on the market. Purdue Pharma, the drug’s producer, recalled it due to the possibility of significant side effects when the medication was used in combination with alcohol. Because apparently, some people need a drink to go with their potent narcotic pain medication. This combination would release hydromorphone, triggering a high drug level in the body that could depress or halt respiration, resulting in coma and even death.
OMNIFLOX, 1992
 https://apnews.com/article/d99b2441ac5190fa107aab2db9db9179
https://apnews.com/article/d99b2441ac5190fa107aab2db9db9179 Omniflox was on the market for about a year before it was banned in 1992. The FDA recalled the antibiotic, also known as Temafloxacin and distributed by Abbott Laboratories, following at least 50 reports of severe responses to the medicine, including low blood sugar, kidney malfunction, and three fatalities. There were also several instances of hemolytic anaemia (the destruction of red blood cells), other blood cell abnormalities in those who had used the medication, and liver dysfunction. Approximately half of the patients who reported kidney difficulties had to undergo renal dialysis treatment.
BROMFENAC (DURACT), 1998
 https://www.washingtonpost.com/archive/politics/1998/06/23/painkiller-taken-off-market-after-deaths/a6188447-39d0-4ae3-b7a7-d553853dffdf/
https://www.washingtonpost.com/archive/politics/1998/06/23/painkiller-taken-off-market-after-deaths/a6188447-39d0-4ae3-b7a7-d553853dffdf/ Duract was an effective painkiller, but it resulted in four deaths, eight liver transplants, and 12 cases of serious liver damage in the 12 months it was available on the shelves. Apart from the more severe side effects, among the 200,000 to 400,000 people who took Duract for extended periods, several victims developed jaundice and severe hepatitis. The medicine was only supposed to be used for 10 days, but patients were frequently prescribed higher doses. All occurrences of death and liver damage occurred in patients who used the medicine for more than 10 consecutive days. There were around 2.5 million prescriptions written for it.
ABLE LABORATORIES DRUGS, 2005
 https://www.nbcnews.com/id/wbna8050556
https://www.nbcnews.com/id/wbna8050556 Sometimes you have to applaud these pharmaceutical manufacturers for the things they can achieve. Due to concerns about the manufacturing process’s quality, all of Able Laboratories’ products were recalled from the market on May 23, 2005. That’s right. All of them. Some drugs were found to be too strong, while others weren’t strong enough. Furthermore, four of its executives were discovered to have fraudulently distributed misbranded and contaminated medications. The recall effectively put Able Labs out of business.
PERMAX (PERGOLIDE), 2007
 https://www.drug-injury.com/druginjurycom/2007/08/permax-recall-c.html
https://www.drug-injury.com/druginjurycom/2007/08/permax-recall-c.html Valeant’s Permax, intended to treat Parkinson’s disease, was on the market for 19 years until 2007. The drug was recalled as it was found to cause a serious heart condition. Valve regurgitation (a condition in which the valves can’t seal tightly, allowing blood to flow back over the valve) causes shortness of breath, exhaustion, and heart palpitations in the mitral, tricuspid, and aortic heart valves. One would think that would be the end of it. However, today, Permax is still available for veterinary treatment in the United States, specifically for pituitary pars intermedia hyperplasia or equine Cushing’s Syndrome (ECS). Showing that drug manufacturers can and will find a way.
MYLOTARG (GEMTUZUMAB OZOGAMICIN), 2010
 https://www.fiercepharma.com/pharma/fda-pfizer-voluntarily-withdraws-cancer-treatment-mylotarg-from-u-s-market
https://www.fiercepharma.com/pharma/fda-pfizer-voluntarily-withdraws-cancer-treatment-mylotarg-from-u-s-market You’d assume that having cancer would get you the best treatment possible. Or, at the very least, treatment that does not shorten your life or cause additional fatal illnesses. Unfortunately, the opposite is often true. Mylotarg was approved by the FDA under the rapid approval program in May 2000 to treat cancer patients with recurrent AML unsuitable for other therapy forms. The trial terminated early because patients receiving Mylotarg died at a higher rate than those receiving chemotherapy alone. To make matters worse, Mylotarg caused a dangerous liver disorder known as a veno-occlusive disease in many of the cancer patients, which is fatal in and of itself. Until 2010, the medicine has been on the market for ten years.
DARVON - DARVOCET, 2010
 http://edition.cnn.com/2010/HEALTH/11/19/fda.removes.drug/index.html
http://edition.cnn.com/2010/HEALTH/11/19/fda.removes.drug/index.html Propoxyphene, a painkiller used by over 10 million people and supplied as a standalone pill under the brand name Darvon as well as as a component of other pharmaceuticals such as Darvocet, was pulled off the market in 2010 after it was connected to potentially fatal cardiac rhythm abnormalities. These heart rhythm abnormalities were not only severe but frequently fatal. Xanodyne Pharmaceuticals Inc. filed the first petition against the medicine in 1978. The FDA asked the drug’s generic manufacturer to follow Xanodyne’s lead. Darvocet was once one of the most widely used pain relievers in Europe and the United States. Today it is entirely prohibited in the United States and the United Kingdom.
TROGLITAZONE, 2000
 https://www.sciencedirect.com/topics/medicine-and-dentistry/troglitazone#:~:text=However%2C%20troglitazone%20was%20removed%20from,because%20of%20possible%20hepatic%20toxicity
https://www.sciencedirect.com/topics/medicine-and-dentistry/troglitazone#:~:text=However%2C%20troglitazone%20was%20removed%20from,because%20of%20possible%20hepatic%20toxicity The Rezulin saga revealed troubling events surrounding the drug’s withdrawal from the market. The FDA’s handling of the situation was also questioned as the FDA apparently delayed the process due to objections from Rezulin’s manufacturer. Additionally, an FDA doctor who voiced concerns about the medicine was subsequently removed from his position at the request of Warner-Lambert (now Pfizer), which caused even further delays in the recall process. Rezulin, an anti-diabetic and anti-inflammatory medication, was found to be associated with hepatitis. The use of the drug resulted in at least 63 deaths and multiple cases of liver failure. Before the recall, Warner-Lambert generated over $2.1 billion in sales from Rezulin.
POSICOR, 1998
 https://www.ahajournals.org/doi/full/10.1161/01.CIR.98.9.831
https://www.ahajournals.org/doi/full/10.1161/01.CIR.98.9.831 In 1998, Posicor, a prescription medicine used for treating hypertension, was recalled, giving us a spectacular example of what can happen when drugs get rushed to market. 123 People died as a result of taking the medication – which, while small compared to other drugs on this list – represents a significant proportion of the 200,000 people who took Posicor in its first year on the market. While the medication is considered safe on its own, it has been discovered to be potentially fatal when combined with at least 25 other drugs.
BAYCOL (CERIVASTATIN), 2001
 https://abcnews.go.com/Health/story?id=117293&page=1
https://abcnews.go.com/Health/story?id=117293&page=1 Baycol was on shelves for four years, during which time it was a regularly prescribed medication for high cholesterol. However, the medicine was discovered to be exceptionally dangerous since it was linked to a very terrible muscle disease. Rhabdomyolysis is a disorder that causes muscle tissue to die and can eventually lead to kidney failure. Before the medicine was recalled, an estimated one hundred thousand people died. The manufacturer, Bayer, paid approximately $1.2 billion in legal damages.
PEMOLINE (CYLERT), 2010
 https://www.nbcnews.com/health/health-news/fda-withdraws-approval-adhd-drug-flna1c9437384
https://www.nbcnews.com/health/health-news/fda-withdraws-approval-adhd-drug-flna1c9437384 On January 27, 1975, the FDA approved Pemoline, a central nervous system stimulant, for treating ADHD. However, reports of liver complications arose in clinical studies in the United States long before its approval. The earliest instances of liver damage were discovered in 1973 and 1974 trials, where fatty liver deposits were found during a biopsy, and liver damage decreased when therapy was stopped but resurfaced when the drug was resumed. Using data from this and earlier investigations, the authors calculated that drug-induced liver enzyme abnormalities occurred in 1% to 2% of Pemoline patients. Cylert eventually got a boxed warning from the FDA in 1999, alerting doctors and patients to the risk of liver damage, but it was 30 years too late. A lot of children developed acute liver failure as a result of Pemoline and required liver transplants. The more unfortunate ones died as a result of the medication.
VALDECOXIB (BEXTRA), 2005
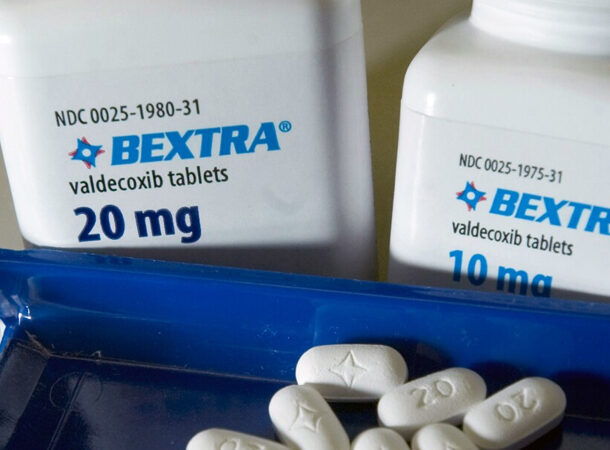 https://abcnews.go.com/Health/PainManagement/story?id=649174#:~:text=April%207%2C%202005%20%E2%80%94,potentially%20life%2Dthreatening%20skin%20reactions.
https://abcnews.go.com/Health/PainManagement/story?id=649174#:~:text=April%207%2C%202005%20%E2%80%94,potentially%20life%2Dthreatening%20skin%20reactions. Bextra, like Vioxx, is a nonsteroidal anti-inflammatory medication used to treat arthritis and other inflammatory conditions. It was taken off the market shortly after Vioxx due to similar concerns – an increased heart attack and stroke risk. In some cases, it was also shown to trigger a deadly skin condition. Despite being less recognized and the subject of fewer lawsuits, Bextra resulted in legal awards of over $2 billion against Pfizer and its subsidiary Pharmacia & UpJohn – one of the biggest criminal fines ever issued in the United States. Oh yes, did we mention the drug also caused an elevated risk for internal bleeding?
FEN-PHEN, 1997
 https://corporate.findlaw.com/litigation-disputes/what-s-the-skinny-on-fen-phen.html
https://corporate.findlaw.com/litigation-disputes/what-s-the-skinny-on-fen-phen.html Fenfluramine/phentermine, also known as Fen-Phen, used to be one of many “miracle” weight-loss medications on the market. In the 1970s, Wyeth manufactured and marketed it as fenfluramine but had limited sales. Then Wyeth introduced phentermine, and everything changed. The drug’s popularity skyrocketed in the 1990s due to intensive ad campaigns, and over 6.5 million people purchased Fen-Phen for weight loss purposes. However, extensive usage of Fen-Phen led to reports of significant lung issues, notably pulmonary hypertension. Following the death of a young woman after taking Fen-Phen for a month, the FDA recalled the medicine, and it was later found that a Wyeth official highlighted concerns about undercounting pulmonary hypertension patients as early as 1994. Over 50,000 victims filed lawsuits against Wyeth, and the firm has lost an estimated $21 billion in legal defences and Fen-Phen damages.
DIETHYLSTILBESTROL (DES), 1971
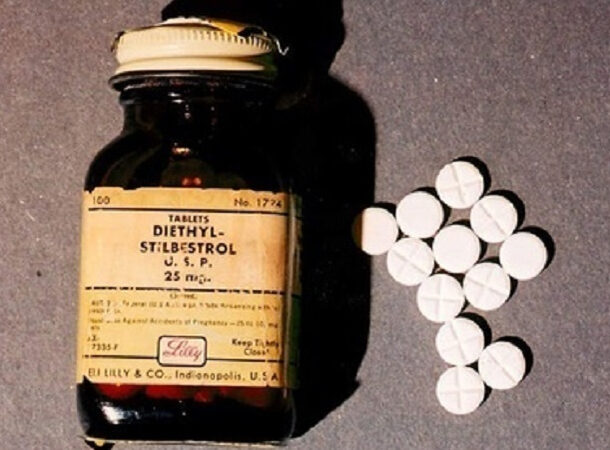 https://www.cancer.gov/about-cancer/causes-prevention/risk/hormones/des-fact-sheet
https://www.cancer.gov/about-cancer/causes-prevention/risk/hormones/des-fact-sheet Diethylstilbestrol was recommended to pregnant women for nearly three decades to minimize the risk of miscarriages and other pregnancy issues. The medical establishment at the time thought that some women did not produce enough estrogen for safe delivery and that DES may compensate for this. Even though research had demonstrated that DES was ineffective in 1953, doctors would continue to prescribe it for years to come. To make matters worse, DES caused a rare form of vaginal cancer in girls exposed to it while still in the womb. Those who suffered side effects from DES won a landmark product liability case in 1980. The California Supreme Court ordered all DES manufacturers to pay an amount in line with their earnings of the drug’s market share while it was being sold as part of the settlement.
PHENYLPROPANOLAMINE (PPA), 2000
 https://www.sciencedirect.com/topics/medicine-and-dentistry/phenylpropanolamine#:~:text=Before%20its%20removal%20from%20the,from%20OTC%20sales%20in%202005.
https://www.sciencedirect.com/topics/medicine-and-dentistry/phenylpropanolamine#:~:text=Before%20its%20removal%20from%20the,from%20OTC%20sales%20in%202005. PPA, a psychoactive drug with a long history of use, makes it to number three on today’s list because the FDA never formally approved it. That’s right. The drug was used for everything from dieting to cold medication to treating psychological illnesses for decades while residing in a kind of limbo where it was neither banned nor endorsed. That is until a study by Yale University in 2000 revealed a link between the drug and cardiac events and stroke, particularly among women. The study indicated that for every 107,000 to 3,268,000 women who used items containing phenylpropanolamine within a three-day period, 1 woman would have a stroke. The FDA believes that if PPA had been replaced with something less dangerous, 200 to 500 strokes may have been avoided each year. Over 25,000 lawsuits have been filed against various drug manufacturers, with judgements for damages totalling hundreds of millions of dollars.
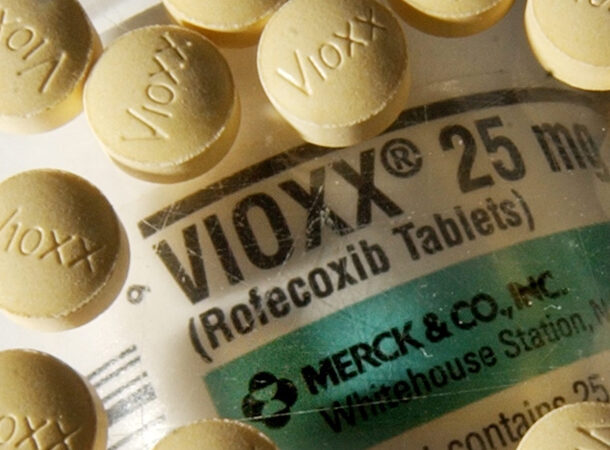
VIOXX, 2004
 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1779871/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1779871/ Vioxx, also known as Rofecoxib, was a medication for pain relief frequently employed to alleviate arthritis pain. At its peak, more than 20 million people used the medicine, earning Merck & Co. $2.5 billion in annual sales. Then, researchers discovered that Vioxx raised the risk of having a heart attack or a stroke. According to one Vioxx report, up to 140,000 patients developed coronary heart disease after using it. In 2004, Merck voluntarily recalled Vioxx. However, subsequent reports indicated that Merck and the FDA had overlooked proof of severe side effects for years before the recall. Merck eventually had to pay $4.8 billion to settle all Vioxx-related litigation. The Vioxx recall is regarded as one of the largest in US history
THALIDOMIDE, 1961
 https://www.understandinganimalresearch.org.uk/news/sixty-years-on-the-history-of-the-thalidomide-tragedy
https://www.understandinganimalresearch.org.uk/news/sixty-years-on-the-history-of-the-thalidomide-tragedy Only one drug can feature in our number-one spot today, as its recall was one of the most transformative recalls in the history of the FDA. Thalidomide, also known as Immunoprin, was developed by Chemie Grünenthal in 1954 and was initially used as a sedative. It was eventually prescribed to pregnant women to alleviate morning sickness. Throughout the late 1950s, thalidomide became available as an over-the-counter medicine worldwide. However, the drug was discovered to have caused thousands of severe birth defects. The National Institutes of Health said it was the “biggest man-made medical disaster ever.”
Following the crisis, Congress enacted the Kefauver-Harris Drug Amendments, which gave the FDA the authority to monitor medications and required manufacturers to document the adverse side effects of their products.



























